In-process QC: Rapid Quality and Stability Testing
You do the science. You focus on your cutting edge research, drug discovery, gene editing, cellular manufacturing etc.
Let Cellected carry out the in-process QC – in line with the ISSCR Standards – with its RIQUEST service.

Cellected’s RIQUEST: a two-stage QC process
RIQUEST – Stage 1:
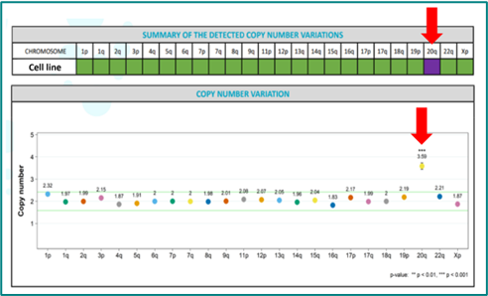
Rapid genomic assessment (3-5 days) to detect over 92% of recurrent genomic abnormalities in key hotspots for human iPSCs, including 20q11.21 and common changes such as 1q, 12p, 17q and others.
For most cultures (~75%) the genomic assessment will be normal and cells proceed to Stage 2.
In the case of a genomic abnormality the cells can now be rescued by our novel RESCUE service

RIQUEST – Stage 2:
Further, advanced cell quality assessments in-line with ISSCR standards:
- health, viability, morphology
- AI-based growth rate and population doubling
- ICC/IF to assess expression of key undifferentiated markers
- process takes less than 2 weeks
Full reporting following the ISSCR standards ensuring the data are ‘publication ready’ and compliant
